by Ryan A. Hall, originally presented at the 2003 SuperSlow Medical Resource Symposium
Introduction
Scientifically, stress is defined as the nonspecific responses of an organism to any demand made upon it (Selye, 1974). Viewed in this light, exercise is nothing more than a physical stressor imposed upon the body to produce an adaptive response. The extent of the adaptation is dependent upon balancing the severity of the stressor (intensity, frequency, and duration) with an adequate recovery interval. Exercise does not produce the adaptation directly. Rather, it serves as a stimulus for the body to produce the adaptive response.
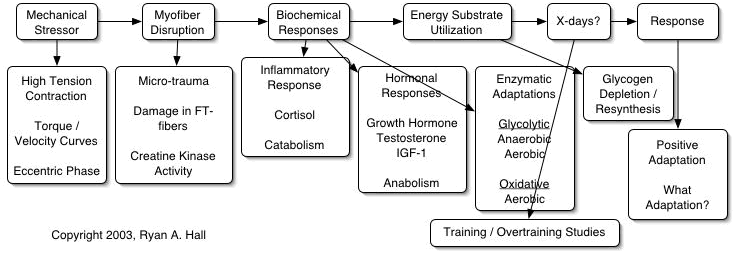
McGuff (1997) has outlined this process in the following schematic:
Stimulus > Organism > X-days > Response
In this model, the stimulus (exercise) is imposed upon the organism (human body), followed by a recovery period (x-days) in order to produce a response (increase in muscular strength and hypertrophy). Moreover, further questions arise from this model. What are the mechanisms of the stimulus? What effect does the stimulus have on the organism? What occurs during the recovery period (x-days)? How much recovery time is required? What happens if the recovery interval is interrupted? Does the recovery interval change with alterations in the stimulus or organism? What is the desired response?
Most of these questions can be answered by looking at existing physiology and medical research literature. A multitude of data is available concerning exercise stress and recovery. This presentation will review research on many aspects of the stimulus and recovery process of skeletal muscle in order to develop a biochemical adaptation model. When viewed together, this data supports the attached biochemical model (Fig 1) of the stimulus and growth process of skeletal muscle.
Following the model, it can be seen that the initial stimulus is a mechanical stressor to the muscle tissue caused by high-tension, low-velocity contractions, occurring during the eccentric (negative or lowering) phase of an exercise (Armstrong RB, Warren GL, Warren JA, 1991). The high-tension eccentric contractions cause disruption or micro-trauma of the myofilaments (contractile proteins), and cytoskeleton of the muscle fibers, usually in the largest fast glycolytic fibers (Friden J, Sjostrom M, Ekblom B, 1983). This initial mechanical damage is followed by an inflammatory response resulting in further protein turnover. Serum levels of creatine kinase are used as a measure of protein damage / turnover. Depending upon the severity of myofiber disruption, serum creatine kinase can increase to very high levels over the next five days, and not return to baseline for 10 or more days (Pedersen BK, Ostrowski K, Rohde T, Bruunsgaard H, 1998). This process must be left uninterrupted or protein synthesis and regeneration of the damaged fibers will not be complete. If an adequate recovery interval is allowed, the body will enter an anabolic state and the disrupted fibers will enhance their composition of contractile protein by increasing the number of myofibrils within the muscle fibers. The previously disrupted fibers will hypertrophy, increasing the resistance to further damage at similar intensities (Armstrong et al., 1991).
However, if another stimulus (workout) is introduced before recovery is complete, a cascade of negative biochemical reactions occurs in the body. Protein turnover will be incomplete, thus causing further disruption to previously damaged tissue. This imbalance between stimulus and recovery leads to an overstressed condition and an increase in the production of cortisol, a major catabolic hormone. An overabundance of cortisol upsets the balance between catabolism (breakdown of tissue) and anabolism (build-up of tissue), favoring the catabolic process (Urhausen A, Gabriel H, Kindermann W, 1995). If this occurs, the organism is now in a chronic state of degeneration called overtraining. Overtrained individuals experience a wide range of conditions, including: muscle soreness / stiffness, tendonitis, suppressed immune system, increased frequency of upper respiratory tract infections, depression, lethargy, weakness, reductions in testosterone, greatly reduced sperm count in men, depressed muscle glycogen reserves, insomnia, decreased exercise performance, and symptoms of Cushing’s disease (Budgett R, 1990; Fry RW, Morton AR, Keast D, 1991). Research indicates that chronically overtrained individuals may require up to three to six months to fully recover after cessation of training (Kuipers H, Keizer HA, 1988). At this point, further results from exercise are not possible.
Results from exercise are dependent upon the proper manipulation of stimulus and recovery. Research indicates that high intensity exercise may require an extended recovery interval. Further evidence shows that as the intensity of exercise increases, greater micro-trauma accumulates, requiring a greater recovery interval (Ploutz-Snyder LL, Tesch PA, Dudley GA, 1998). Details of the biochemical model and application to exercise prescription will be covered in the presentation and in report format at a later date.
Copyright 2003, Ryan A. Hall.
References:
Armstrong RB, Warren GL, Warren JA. Mechanisms of exercise-induced muscle fibre injury. Sports Med 1991 Sep;12(3):184-207.
Budgett R. Overtraining syndrome. Br J Sports Med 1990 Dec;24(4):231-236.
Friden J, Sjostrom M, Ekblom B. Myofibrillar damage following intense eccentric exercise in man. Int J Sports Med 1983 Aug;4(3):170-6.
Kuipers H, Keizer HA. Overtraining in elite athletes: Review and directions for the future. Sports Med 1988 Aug;6(2):79-92.
McGuff, D. The dose-response relationship of exercise. Ultimate Exercise: Bulletin Number 1, 1998.
Pedersen BK, Ostrowski K, Rohde T, Bruunsgaard H. The cytokine response to strenuous exercise. Can J Physiol Pharmacol 1998 May;76(5):505-11.
Ploutz-Snyder LL, Tesch PA, Dudley GA. Increased vulnerability to eccentric exercise-induced dysfunction and muscle injury after concentric training. Arch Phys Med Rehabil 1998 Jan;79(1):58-61.
Selye, Hans. Stress Without Distress. Signet, New York, 1974.

Comments on this entry are closed.
“Serum levels of creatine kinase are used as a measure of protein damage / turnover. Depending upon the severity of myofiber disruption, serum creatine kinase can increase to very high levels over the next five days, and not return to baseline for 10 or more days (Pedersen BK, Ostrowski K, Rohde T, Bruunsgaard H, 1998). This process must be left uninterrupted or protein synthesis and regeneration of the damaged fibers will not be complete. If an adequate recovery interval is allowed, the body will enter an anabolic state and the disrupted fibers will enhance their composition of contractile protein by increasing the number of myofibrils within the muscle fibers. The previously disrupted fibers will hypertrophy, increasing the resistance to further damage at similar intensities (Armstrong et al., 1991).”
This seems to support the BBS protocol of training the full body only once per week (every 5-10 days for most people). Has further research come out that condradicts or disproves this conclusion? I ask this because I believe you have now switched to two workouts per week for yourself, and many other HIT advocates recommend 3 workouts per week.
Thanks for hosting such a great website and continuing to provide top-notch training information!
Hey Roger,
Like most things, this depends on the individual, and some people may recover more quickly and others more slowly. The only way for most of us to practically determine this is to track and compare workout performance and body measurements over time and to experiment. Twice weekly seems to be a good starting point for most people, but there are some people who require a lot less frequency (at least per muscle group) to recover.
Thanks for your response, Drew. I understand that recovery time from HIT varies between different individuals, with some people recovering quicker and others slower. However, if I understand the study cited by Ryan Hall correctly, the variability of “serum creatine kinase increase” (which measures protein damage/turnover) ranges between “5 and 10 days or more” before returning to baseline, indicating that the muscle fibers are ready for further stimulus.
If that’s the case, then performing a HIT routine more frequently than every 5th day would seem counterproductive, unless other studies have demonstrated that the serum creatine kinase increase returns to baseline in less than 5 days in some people. Are you aware of any studies that show this? Or are you basing your conclusion that working out “twice weekly” is a good starting point “for most people” more from practical experience training yourself and others? Thanks again for your input.
Hey Roger,
If the goal is muscular strength and size increases it is more useful to look at studies that directly measure this, rather than indirect factors in isolation which may not give a complete picture. If this really was the case people would be losing strength training more frequently, but I just don’t see that happening with most people, either in research or my own experience as a trainer.
“If the goal is muscular strength and size increases it is more useful to look at studies that directly measure this, rather than indirect factors in isolation which may not give a complete picture.”
Yeah, I see your point, and you may be correct about that. But how many studies on strength and hypertrophy have been done using a full-body HIT protocol that measures training volume and frequency over an adequate time-frame of 90-180 days or longer, and with a large enough group of participants to be statistically relevant? It seems like most studies are so poorly constructed as to be practically worthless.
“If this really was the case people would be losing strength training more frequently, but I just don’t see that happening with most people, either in research or my own experience as a trainer.”
I don’t question your experience in the least, but Ryan Hall seems to have experienced the exact opposite as a trainer…at least as it relates to “advanced” subjects (which I assume would be those who have at least 6 months of more of proper HIT training under their belt?). This is from his blog posted on 8/2/14:
“With advanced subjects, the biggest issue I have is modulating the recovery interval. For several years now, also depending upon the client’s consistency, I’ve been moving those individuals to a two way split routine, still maintaining a once per week schedule. Although, I do have very few clients that can still maintain a twice per week schedule with a two way split. The two way split routine consists of (1) an upper body day, and (2) a lower body with core/spine/neck work. For extremely advanced subjects/individuals requiring an extended recovery interval, I have been experimenting with a three way split routine, consisting of (1) upper body, (2) lower body/legs, (3) core/spine/stretch.” (http://www.exercisesciencellc.com/blog/files/47f1a2425fcac966c245714718bbaf53-5.html)
Do you two simply disagree about this? Or am I reading more into it than is warranted?
Hey Roger,
I can only comment on my experience, and although I’ve had many clients who have needed to cut back to training once weekly the majority have done better training more frequently. There have been numerous instances where clients have had to cut back from twice to once weekly over the years due to either scheduling conflicts or a lack of finances, and in the majority of cases their progress slowed (comparing over time, not workout to workout). Some studies have even shown better results training three times a week than twice weekly (Braith RW, Graves JE, Pollock ML, Leggett SL, Carpenter DM, Colvin AB (1989). Comparison of 2 vs 3 days/week of variable resistance training during 10- and 18-week programs. Int J Sports Med. 10(6):450-4.) and plenty of studies supporting the recommendation of training three days a week for beginners and two for advanced trainees (Rhea MR, Alvar BA, Burkett LN, Ball SD (2003). A meta-analysis to determine the dose response for strength development. Med Sci Sports Exerc. 35(3):456-64.).
The most important thing to keep in mind is this varies considerably between individuals, and any training volume and frequency should be considered a starting point from which to make adjustments based on individual response. If you’re doing everything else right and not progressing on two full-body workouts per week, you could cut back to three workouts every two weeks or go to an upper-body/lower-body split like Ryan is using and compare results. You have to experiment, keep accurate records of what you’re doing and how it’s affecting goal-specific measurements, and adjust accordingly. It is also important to keep in mind, however, that progress will slow over time and it is unrealistic to be able to expect to regularly add five to ten pounds to exercises, and I suspect that many people cut back their volume and frequency too much as they advance when what they ought to be doing is reducing the weight increments.
Hi Drew,
Thanks again for your response. The studies you linked to are interesting, but I’m not sure how much practical relevance they have to a properly done HIT program. The first one used sedentary untrained men and women in their late twenties, performing one set of leg extensions to failure for seven to ten reps, for a period of ten to eighteen weeks. Would it have produced the same results using a broader range of age groups? Or using squats or leg presses rather than an isolation exercise? Or using a “Big Five” type of routine done to MMF? Perhaps I just have terrible recovery ability, but I can’t imagine doing the “Big Five” routine intensely 2 or 3 times a week for very long without completely burning out, even when I was in my twenties. I remember doing a twenty-rep breathing squat routine 2 times per week for ten weeks when I was about 25 years old, and I’m positive that I was totally over-trained by the end of it. I made it up to 300 lbs for twenty reps at a body weight of about 160 lbs, but I bet I would have progressed quite a bit beyond that point if I was only doing it once per week.
The second study was a meta-analysis of 140 different studies, so who knows how well or poorly each study was set up and conducted. It seems like there would be way too many variables between the studies to draw any type of relevant conclusion from such a meta-analysis. It states that “four sets per muscle group elicited maximal gains in both trained and untrained individuals,” so I highly doubt that very many of those studies were analyzing properly done full-body HIT routines.
Anyway, I agree with you “that progress will slow over time and it is unrealistic to be able to expect to regularly add five to ten pounds to exercises.” Whether too many trainees “cut back their volume and frequency too much as they advance when what they ought to be doing is reducing the weight increments” is an open question for me. I just finished reading “High Intensity Training the Mike Mentzer Way,” and he came to the opposite conclusion as a personal trainer, sometimes cutting his advanced trainees back to a 3 way split done once every 5-14 days or more before they started to resume progress. That seems to line up with Ryan’s experience too. So who knows? I certainly don’t have the answer. I can’t imagine only hitting the gym once every 14 days or so though, so once per week is probably the most I’ll ever cut back to.
Thanks again for your input, and best of luck at the upcoming 21 Convention!
Roger,
The first shows why two times weekly is a good starting point, and while the meta analysis may be off (Rhea has gotten things pretty wrong before) all of this reflects what I’ve experienced training hundreds of people over a period of twenty years. In most cases where a client has reduced frequency due to scheduling or financial reasons they made about the same amount of improvement each workout, but their rate of progress over time would be halved. Some people who had difficulty progressing at twice weekly have done better cutting back to once a week, but the majority make pretty good progress training twice weekly.
By the way, “reducing the weight increments” is definitely a good idea as one advances toward their genetic potential, regardless of the workout frequency chosen. I’m a big fan of using “micro” plates and adding as little weight as possible once I start getting stuck at a particular weight. That should probably be tried before reducing training frequency to more than once per week…
Hi Drew,
So, would you disagree with Ryan Hall’s comments, here:
“Usually, when a particular twice per week subject reduced the frequency of training to once per week, strength increases were almost immediate and continuous. From my recollection, this occurred in approximately 97% of the original subjects. Rarely did the reverse occur, although, there were a small percentage of subjects who progressed less when transitioning to the once per week schedule.” (http://www.exercisesciencellc.com/articles/results_curve/results_curve.html)
By the way, just to be clear, I highly respect your experience and opinion on these matters, so please don’t take my comments here in any other way. I’ve also emailed these same questions to Ryan Hall (who hasn’t responded yet), as I’m interested to hear his perspective as well. As I said before, the answer to how this applies to “most people” is still an open question for me. I’ve already determined that a full-body HIT routine once every 5th or 7th day is about the most I can handle. I’m currently doing my workout once every 7th day simply because it’s more convenient to hit the gym on the same day every week for my work schedule. This is my current routine:
Workout A:
Machine Bench Press
Reverse Grip Pulldown
Machine Lateral Raise
Triceps Machine
Machine Preacher Curl
Leg Press
Standing Calf Raise
Machine Crunch
Workout B:
Machine Overhead Press
Machine Row
Pec-Deck
Triceps Pressdowns
Cable Curl
Trap-Bar Deadlift
Standing Calf Raise
Machine Crunch
Thanks again for your input!
Roger
Roger,
I’m not disagreeing with Ryan and have seen the same thing in some clients, I just have not found this to be the case with nearly as large a percentage. Also, it is important to consider that progress needs to be looked at over time, not on a workout to workout basis. Some people may make better improvements from workout to workout training less frequently, but when cutting frequency in half unless their rate of improvement between workouts at least doubles, their overall progress will be slower.
If halving the frequency doubles the rate of progress between workouts, the net progress would be the same but the reduced frequency would be superior because of the greater results relative to the time invested and reduced stress on the body. If halving the frequency does not at least double the rate of progress, however, then you have to look at what is more important to the individual, progress over time or return on time invested. If halving the frequency only resulted in fifty percent greater improvement between workouts it would result in slower progress over time but a better return on the time invested.
Unless it is for time efficiency, scheduling, or financial reasons, the only time I would recommend cutting back would be when progress is very slow (taking into consideration how advanced the trainee is) or has stopped completely despite everything else being in order (diet, sleep, stress management, etc.), but usually only after first trying an upper-body/lower-body split or a push/pull/legs split (usually using the splits in High Intensity Workouts).
To determine this I recommend taking up to two weeks off of training completely, then resuming at the reduced frequency for several weeks and comparing. If someone makes better average progress over the course of a month on the reduced frequency, they should continue with it.